Chemists are Artists
|
In this project we were given the opportunity to make artwork using chemistry. The artwork created would be put in the new STEM building at San Marin (right), so it was very important to insure that our artwork would be high-quality, captivating and informative. We were given a lot of freedom on the content of our artwork, so it took a lot of brainstorming to come up with our plan.
My group and I decided that making a piece that brought awareness to a world issue could be really interesting. We decided to highlight pollution and global warming, since that is a growing issue that isn't being solved. To do so, we wanted to create a burnt-looking art piece. We thought that by burning wood, we could not only make a point towards our message but also utilize the amazing chemistry that goes into making substances burn. Once we had a basic consensus on what we wanted to convey, we went to researching ways to go about it. We eventually came across burning Ammonium Chloride. Our entire process is shown in the sideshow below. |
Project Requirements:
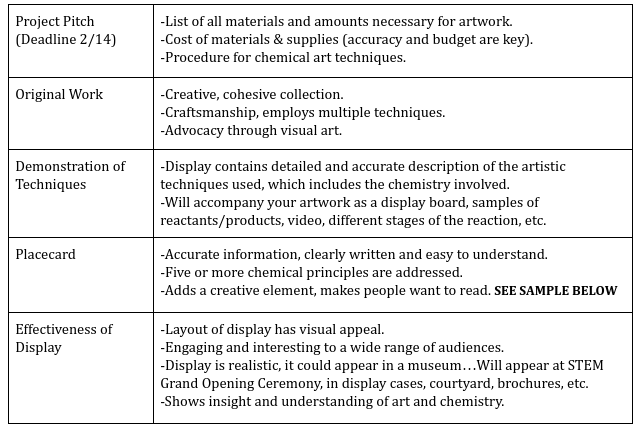
The Driving Question for this project was: "How can we use chemistry to design a work of art for our new STEM building?" and we were given this Prompt:
|
Above is the requirements we were given for this project. From the beginning it was obvious that this was going to be an extensive project, especially because the outcome would be displayed for future students. This was not a setback, though, we just used it as motivation to work harder and create a better project.
|
"All artists are chemists. Artists understand and study the properties of specific materials and find ways to explore these properties to express views of themselves and the world around them. Art is the result of the human need to express ourselves. It tells stories of societies, eras, and individuals. Your challenge is to create a work of art that will be part of a four-piece cohesive collection to be displayed in our new STEM Science building.
Throughout this unit we will focus on the different types of chemical reactions but it will be your task to incorporate this into a creative and timeless piece of art. The overarching theme is that chemistry is about change. Intertwined through each activity will be discussions of these changes of chemical interactions. It will be important to focus on the observable properties of the materials before and after the reaction and the atomic level explanation of what is occurring during a chemical reaction." |
Our Message:
As I said before, our message was to spread awareness to pollution and global warming. We created four pieces that highlighted this message. The first piece was about the pollution that comes with cities, factories and industrialization. To portray this, the image of a smoking factory is burnt into the wood. The second piece is about ocean pollution, and shows sea creatures swimming among human litter. The third piece is about deforestation, and shows a tree cut down. The fourth and final plank brings the whole idea together by showing the world on fire to symbolize how humans are ruining where we live. The images of all these pieces are showcased towards the bottom of this page, under The Artwork.
Trial and Error:
|
Our original idea was different from what we actually ended up doing. We originally planned to demonstrate the combustion reaction of lighting gunpowder, but we wanted to do so in a pattern that would make a really cool art piece. The setback for this would be that in order to demonstrate the chemistry, we were required to make the gunpowder. We researched the compound and found that the ingredients we needed were Potassium Nitrate (75%), Charcoal (15%), and Sulfur (10%). Once we had acquired all of these chemicals, we measured them out and ground them together to match the fine consistency of actual gunpowder.
Chemical equation: 10KNO₃ + 8C + 3S ⟶ 2K₂CO₃ + 3K₂SO₄ + 6CO₂ + 5N₂ In theory, this should have worked, but it went wrong when it came to burning. At first, it did not light at all, but once we added more, the entire block of wood lit up in bursts of explosions, following no pattern and emitting a horrid smell. It was after this trial that we decided to switch gears. |
The Chemistry:
When we switched ideas, we wanted to keep the same general theme. Since the gunpowder art didn't go as planned, we went to researching reactions that would have similar results. Eventually we stumbled upon ammonium chloride, which is a clear chemical that chars when heat is applied. This would leave a burnt look on the wood, but only in places that the ammonium chloride was painted on.
The reaction that takes place when ammonium chloride encounters heat is a decomposition reaction.
The chemical equation is: NH₄C ⟶ NH₃+HCl
The reaction that takes place when ammonium chloride encounters heat is a decomposition reaction.
The chemical equation is: NH₄C ⟶ NH₃+HCl
|
To the right is an example of the chemical reaction taking place.
More Terms to Learn:
|
|
The Artwork:
|
To the left is a photo of the final art pieces that my group and I completed. These art pieces make a statement about pollution and global warming, and were made with a reaction between heat and ammonium chloride. In the STEM building, these pieces would be framed together in this way, with the place card in the space between them.
The plank on top signifies deforestation by showing a cut down tree. To the left is a plank that showcases human pollution from urban places. To the right is a piece about ocean pollution, and below is a piece that stands for global warming. |
Placecard:
|
One of the main requirements for this project was to create a place card. This card was to be displayed with the art piece that was created. My group and I decided that we should just create one place card for all four art pieces because it was a very cohesive project, and that place card is shown to the right.
The directions for the place card were pretty specific, but also gave creative freedom. The first requirement was to put our names so viewers could see who created each piece. The second was a title of the collection, which was "Earth on Fire". It was also supposed to show the chemistry and look professional. The overall goal of these place cards were to let the viewers know that the art was created by chemistry, and to do so, the place card had to catch people's eye. We used colors and bold lettering to tie the place card together and ensure that attention was drawn to it. |
More about reactions:
Balancing Chemical Equations:
- The first step to balancing a chemical equation would be to identify all of the elements in the equation.
- Keep in mind that once balanced, the number of each atom must be the same on either side of the equation.
- To make them balanced, a coefficient is added before the elements
- Balance the elements one by one, making sure that you change the coefficients and not the subscripts
- The subscripts show the charges for each element, and those need to be balanced at the end as well.
- Balancing can be made easier by creating a table with each element's amount and charge on each side of the equation
- Once the balancing is done, check your work to make sure that everything is balanced.
|
Solubility Guidelines:
The chart to the right was used all throughout this unit in chemistry during work with double displacement reactions. The solubility guidelines show what compounds will be soluble, and with what exceptions. We used this table to predict if a precipitate would be created in a reaction. Although my group did not use this in our art project, others did. Many people created paint, and to do so they created colored precipitates and mixed the products with a paint base. They would use the solubility guidelines because it would allow them to confirm that a precipitate would form in the reaction they created. |
|
Reactivity Series:
This series of metals is in order from most reactive to least reactive, with most reactive on top and least reactive on the bottom. This table (left) is used in single displacement reactions. It is used to determine if a reaction will take place, and which metal will replace which. In a single displacement reaction, metal A will replace another metal B in a solution if A is higher up in the series. |
Reflection:
Two skills that I successfully utilized in this project were critical thinking and leadership. The first skill was critical thinking, which includes creative problem solving, solution finding, and resourcefulness. I feel like my group and I used creative problem solving when our first attempt failed. Although we did not find success at first, we powered through and did extra research to get the results that we wanted. It took extra work and perseverance to get to the place that we did. We also used resourcefulness because in this project, we had to acquire all of the chemicals and pieces needed for our art project, and if those items weren't available, we had to make do with what we had. Another skill that I successfully utilized was leadership. This project had some obvious obstacles, so taking a leadership position was important to ensure productivity. With the six C's, leadership could fall under communication or character. An example of when I used leadership was when the group was trying to brainstorm ideas. I gave an idea for the central theme, and then helped to find chemistry-related art projects that could fall under that central theme. Overall, the two things that I grew in in this project were critical thinking and leadership.
Two skills that I think I could improve upon after this project would be flexibility and open-mindedness. Within the six C's, these skills fall under collaboration. Our group collaborated extremely well, so we eventually obtained the level of flexibility that we needed, but I could definitely improve my level of flexibility for the next project. I think that I often get ideas in my head that I really want to work, so when obstacles come around like what we faced with the makeshift gunpowder, it was hard to get around that original plan to find another one. I think that this also ties into open-mindedness because if I had been open to any possible solution to the first attempt, it could have been a lot easier to solve.
Other than the obstacles that we faced, I think that my group and I did a really good job in this project, and it was a really good learning experience. It was also really interesting to put chemistry into real-world projects like art, rather than just try to conceptualize microscopic things.
Two skills that I think I could improve upon after this project would be flexibility and open-mindedness. Within the six C's, these skills fall under collaboration. Our group collaborated extremely well, so we eventually obtained the level of flexibility that we needed, but I could definitely improve my level of flexibility for the next project. I think that I often get ideas in my head that I really want to work, so when obstacles come around like what we faced with the makeshift gunpowder, it was hard to get around that original plan to find another one. I think that this also ties into open-mindedness because if I had been open to any possible solution to the first attempt, it could have been a lot easier to solve.
Other than the obstacles that we faced, I think that my group and I did a really good job in this project, and it was a really good learning experience. It was also really interesting to put chemistry into real-world projects like art, rather than just try to conceptualize microscopic things.